A drug company has asked the United States Food and Drug Administration (FDA) for permission to sell a birth control medicine “over the counter.” That means it can be bought without providing a doctor’s order, or prescription. HRA Pharma, a French company, sent the FDA a written request Monday. HRA’s hormone-based drug comes in pill form and prevents pregnancy. Such contraceptive medicine was introduced in the 1960’s and has become the most common form of birth control for women in the U.S. But, it has always required a prescription for purchase. The pill can lead to blood clots in some users, although this is rare. HRA’s request to the FDA argues that users can measure the risks on their own and use the pill successfully. “For a product that has been available for the last 50 years, that has been used safely by millions of women, we thought it was time to make it more available,” said Frederique Welgryn, HRA’s chief strategy officer. The HRA medicine is called Opill and is not sold in the U.S. The FDA could approve it for sale by next year. Many women’s health and reproductive rights supporters want other prescription contraceptives and abortion pills to make the move to over-the-counter. The FDA action could guide many other decisions about women’s health care in the years to come. Reproductive freedom is a major issue in the country. Last month, the U.S. Supreme Court overturned Roe v. Wade, the 1973 decision that established the constitutional right to seek medical abortions.
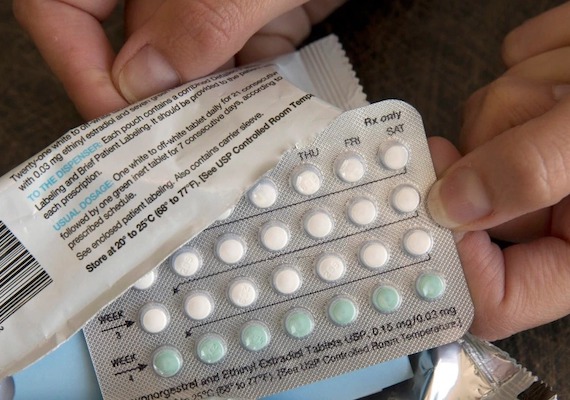
A drug company has asked the United States Food and Drug Administration (FDA) for permission to sell a birth control medicine over the counter.
counter
downer
sounder
cloud
Which of the following is a definition of the word strategy?
a plan of action or policy designed to achieve a major or overall aim
tasting of, containing, or preserved with salt
casual yet attractively stylish
the study of living organisms
When was the contraceptive pill introduced?
1960s
1970s
1940s
1950s